Indirect ELISA, conventional but efficient
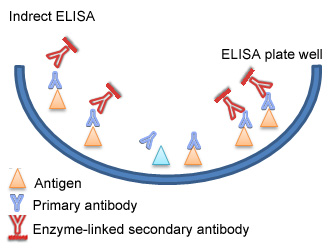
Indirect ELISA is a two-step ELISA which involves two binding process of primary antibody and labeled secondary antibody. The primary antibody is incubated with the antigen followed by the incubation with the secondary antibody. However, this may lead to nonspecific signals because of cross-reaction that the secondary antibody may bring about.
1. Micro-well plates are incubated with antigens, washed up and blocked with BSA.
2. Samples with antibodies are added and washed.
3. Enzyme linked secondary antibody are added and washed.
4. A substrate is added, and enzymes on the antibody elicit a chromogenic or fluorescent signal.
» Learn more about indirect ELISA protocol
Indirect ELISA advantages
- • High sensitivity: More than one labeled antibody is bound per antigen molecule;
- • Flexible: Different primary detection antibodies can be used with a single labeled secondary antibody;
- • Cost-saving: Fewer labeled antibodies are required.
In the indirect ELISA test, the sample antibody is sandwiched between the antigen coated on the plate and an enzyme-labeled, anti-species globulin conjugate. The addition of an enzyme substrate-chromogen reagent causes color to develop. This color is directly proportional to the amount of bound sample antibody. The more antibody present in the sample, the stronger the color development in the test wells. This format of indirect ELISA is suitable for determining total antibody level in samples (Newcastle disease virus, B. abortus, etc.). Detailed information about indirect ELISA application in the determination of antibody titer and procedures of antibody concentration determination are discussed in the following section of ELISA applications.
