ELISpot Schematic Procedure
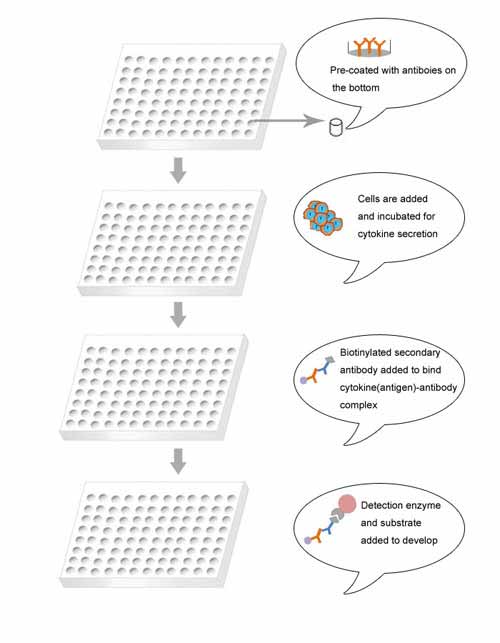
Brief Description
The ELISpot assay was originally designed for the detection and enumeration of the antibody-secreting cells. Subsequently ELISpot has been widely used to assess the cytokine release of various types of cells with or without stimuli. Therefore one significant applications of ELISpot is to evaluate the immune response induced by vaccine. For example, ELISpot technique can be applied in the diagnosis of drug allergy of patients, determining them undergoing benign or severe drug allergic reactions.
ELISpot Assay (IFN-γ)
More specifically, IFN-γ-based assay is the most popular form of the ELISpot technique. IFN-γ is a T cell specific cytokine and dramatically secreted by activated T cells. The IFN-γ ELISpot assay has been used extensively for the quantification of immune responses in vaccine discovery for the prevention and treatment of various diseases like AIDS, tuberculosis.
Due to advantages of high specificity, sensitivity and wide range of detection, ELISpot is perfectly suited and a pivotal tool for clinical trials-development, drug discovery, immunity monitoring, cell-type differentiating, etc.
ELISpot protocol
The buffers:
1. Coating Buffer (1× Phosphate Buffered Saline [PBS]):
8gNaCl; 0.2g KCl; 144g Na2HPO4, 0.24g KH2PO4, H2O to 1 liter Adjust pH to 7.2, autoclave or sterile-filter and store at 4°C
2. Blocking Solution:
Cell culture medium containing 10%Fetal Bovine Serum (FBS) and 1% Penicillin-Streptomycin–L-Glutamine (Gibco)
3. Wash Buffer I:
1×PBS containing 0.05% Tween-20 (0.5ml Tween-20 per 1L PBS)
4. Wash Buffer II:
1×PBS
5. Dilution Buffer:
1×PBS containing 10% FBS
Standard ELISpot Protocol:
- Incubate the plate in room temperature with 100μl of 70% ethanol for 10 min. As for treating the PVDF membranes, the ethanol concentration and incubation time might differ for different experiments and should better to be optimized before addition of capture antibody. If ethanol doesn't work well, try methanol.
- Discard the ethanol and wash with PBS for 3 times.
- Add in appropriate amount of primary antibody solution (with pre-optimized concentration or follwing manual, normally 100μl), cover the top and incubate overnight at 4°C.
- Discard the fluid and wash with PBS.
- Block with 100μl of 12% fat-free milk solution for 2 h at RT.
- Carefully pipet out of the fluid.
- Wash with PBS.
- Add in 100μl of cell suspension (containing appropriate amount of cells and stimuli). Cover the top and incubate at 37°C for 15-20 h. Don't disturb.
- Carefully pipet out of the fluid.
- Incubate with washing buffer of 100μl per well at 4 °C for 10 min.
- Wash with washing buffer for 3 times.
- Enzyme-biotin solution is prepared with 1% BSA diluted in PBS and appropriate amount of enzyme-biotin complex.
- Add in development reagent.
- After about a 5-15 min development, discard the fluid and wash PVDF membrane (edge) with dH2O, carefully dry on blotting paper.
Note: when storage the plate is better to be inverted.
